In re: Elysium Health-ChromaDex Litigation, No. 17-cv-7394 (LJL) (S.D.N.Y. Feb. 11, 2022)
This is the main liability opinion. Because the supplement
facts here are less generalizable than the survey methodologies, I will try to
skip over lots of detail, but a fair amount of the detail is legal. Both
parties get partial wins/losses on summary judgment. The parties make nutritional
products claimed to improve cellular health and cellular aging.
ChromaDex makes Niagen, whose active ingredient is synthetically-produced
isolated nicotinamide riboside (NR). NR is “a naturally occurring form of
Vitamin B3 that increases nicotinamide adenine dinucleotide or NAD+, an
essential coenzyme required for life and cellular functions,” which is “linked”
to cellular health and cellular aging. ChromaDex
used to just supply ingredients to others, but it launched Tru Niagen, a DTC
supplement whose active ingredient is Niagen.
“Elysium was until recently one of ChromaDex’s customers.” Its
primary product is a dietary supplement called Basis, a direct-to-consumer
product which combines NR with a different substance, PT, which also used to be
supplied by ChromaDex. It had other companies manufacture NR on its behalf and
supply it with PT.
An expert panel deemed Niagen generally recognized as safe
(GRAS) as a food additive for “vitamin waters, protein shakes, nutrition bars,
gum and chews,” and ChromaDex submitted their results to the FDA (a voluntary
process). The FDA had no objection (but didn’t make its own determination of
GRAS status). Elysium prepared its own GRAS notification for NR but did not
submit to the FDA, and its GRAS assessment for PT wasn’t reviewed by an
independent panel. The FDA also reviews new dietary ingredients (NDIs); a
supplement with an NDI is deemed adulterated unless it “contains only dietary
ingredients which have been present in the food supply as an article used for
food in a form in which the food has not been chemically altered” or the
manufacturer or distributor of the dietary ingredient or dietary supplement
submits an NDI notification to the FDA with the information it used to conclude
that the supplement is reasonably expected to be safe. ChromaDex submitted NDI
notifications for Niagen, but Elysium had not (yet) for Basis. Elysium has
submitted an investigational new drug application (IND) for Basis for the
prevention of acute kidney injury in surgical cardiac patients; NDI and IND
regulatory processes are different but similar in “scientific rigor around
safety.”
The relevant studies are about whether NR, or the parties’
products, are safe and whether they increase NAD+ in humans.
One of Elysium’s counterclaims was based on statements that
appeared on a blog, Right of Assembly, upwards of 20 times:
ChromaDex isn’t allowed to say that
NR treats any disease, because the FDA has not approved that. But the FDA
doesn’t regulate me, so I am free to tell you that the scientific evidence is
growing that NR supplements replenish cellular NAD, which can protect against
MANY ailments, including [broad list of serious conditions] and in my own
experience, Restless Legs Syndrome (RLS). You can find out more here: [link].
Was this commercial advertising or promotion? Elysium argued that Right of Assembly was “a
marketing website for Tru Niagen for which ChromaDex pays commissions to Shelly
Albaum for Tru Niagen customers referred through the website.”
The court first held that the challenged statement on its
own wasn’t pure commercial speech because it “merely purports to convey
information about NR and NR supplements” and didn’t refer to ChromaDex or its
product; the link was not to a site that itself allowed purchases. Elysium
argued that the website as a whole was a referral website for Tru Niagen, which
advertised Tru Niagen at the top of every page. The court agreed that this was
the right approach in theory. There’s a difference between a statement about
product benefits made by Consumer Reports and the same statement by a
person with an economic motive who also displayed a banner, “Buy This Product.”
However, Elysium didn’t show that all the pages of the blog had a Tru Niagen banner,
only the homepage. [I would have thought that was enough.]
But there was another problem. Elysium didn’t make the
statement. The Lanham Act has been extended to cover statements made by the defendant’s
agent. But “[t]here are no reported cases where Lanham Act liability is
extended to a company that merely benefits from the statement and compensates
the author for the statement in the absence of an agency relationship or
evidence that it has exercised control over or caused the statement.” Did
referral commissions suffice to show an agency relationship? [The FTC 100%
thinks so.] But Elysium didn’t show that ChromaDex controlled Albaum. Its
affiliate program listed him as one of ChromaDex’s affiliates, and Albaum
listed himself [based on pronouns used elsewhere] as one on Right of Assembly,
but that doesn’t show an agency relationship, and questions from another
marketing consultant about “[w]ill she [sic] work with you to ad [sic] content
regularly” supported the inference that Albaum didn’t report to ChromaDex and didn’t
even regularly work with the company in creating content. [This kind of holding
is a disastrous route to claim laundering and doesn’t make the supplement
industry look any better.] Albaum emailed a ChromaDex employee with a different
post that he said he chose not to post but would if “[employee] wants me to”
and requested that the employee “consider these ideas seriously,” and the
employee flagged a ChromaDex citizen petition filing for Albaum. This wasn’t
enough to establish direction or control. Also, Albaum was a shareholder. “That
Albaum was a shareholder of ChromaDex gave him an independent economic incentive
to tout its products no different than the economic incentive that any
investor, whether an activist institutional investor or a retail investor,
might have to promote the products of a company in which it has a financial
interest.” That wasn’t enough to make the shareholder the company’s agent.
Here’s a statement that will probably be quoted widely:
[T]he fact that ChromaDex might
reward Albaum or any other third party for steering business ChromaDex’s way
does not establish that ChromaDex has the ability to exercise any control or
direction over the statements that Albaum might make. A whole industry exists
of social media influencers, who create their own content touting products and
receive commission on sales of those products that stem from their advertising.
A claim might lie directly against such persons whether under federal law or
the state law of trade defamation if they make a false and misleading
statement. But in the absence of evidence that an influencer is making the
statement on behalf of the defendant or at the defendant’s direction or under
its control rather than simply for its own benefit, the company cannot be held
liable on a principal– agent theory.
Since the evidence didn’t show that ChromaDex requested any
particular content, their communications about messaging were not evidence of
agency. ChromaDex didn’t hold Albaum out to be a member of its staff, endorse
his statements, or say that his blog was “correct,” and the blog disclaims such
affiliation. Thus, any false advertising claim would lie against Albaum, not [directly]
against ChromaDex.
Falsity: Elysium won summary judgment on lack of falsity for
many of its statements challenged by ChromaDex, though a uniqueness statement
remains for a factfinder.
Safety/purity claims based on a study, claiming safety/no
serious adverse effects: These were establishment claims and the study was good
enough to support them, so the claims weren’t literally false. The fact that
the study found evidence of increased LDL cholesterol among subjects and said that
further research was needed to fully understand whether these changes were
attributable to Elysium’s Basis did not disprove safety; it didn’t even
establish that Basis caused those changes, nor did the evidence show that the
increase was significant, given that LDL fluctuates day to day in healthy
people within the range showed by the study. Customer questions/complaints about
increased LDL also weren’t enough to show the study unreliable, especially
since that was 41 tickets among over 525,000 messages over four years.
Statements about clinical studies were ok, even though “clinically
proven to raise NAD+ levels” was based on a study that used NR from ChromaDex.
The study was still sufficiently reliable to support Elysium’s claim. “[T]here
is no evidence to suggest that the NR Elysium used after ChromaDex was no
longer its supplier was anything other than materially identical from a
functioning, safety, and efficacy standpoint to the NR that it sourced from
ChromaDex at the time of the study.” Such was the testimony of ChromaDex’s
regulatory consultant; its FDA expert concluded that there were “important
differences in the specifications, solvents, by-products (including acetamide),
and impurity specifications,” but admitted in deposition that he did not know
whether they had any safety implications whatsoever, and offered no opinion at
all about effects on efficacy/functioning. The court was not willing to hold
that “a manufacturer of a product is required, any time it changes the sourcing
of an ingredient in an otherwise-identical formulation, to either abandon any
claims based on its previously-conducted studies or to conduct new studies to
confirm that the change in sourcing did not change the results,” at least where
there was reason to consider the new source “materially identical.”
“Basis is the first and only supplement clinically proven to
increase and sustain NAD+ levels” was also mostly ok because of the and
sustain, which its evidence supported for a chunk of the relevant time
period since the other studies were more short-term. (It was also first to
market.) However, making this statement after a new study purportedly showed ChromaDex’s
product also sustained increased levels could be literally false. There was an
issue of fact whether this study did show that; a jury could therefore find
literal falsity.
What about statements about how a product was “designed”?
Elysium said that its proprietary formulation of two ingredients was “designed”
to work together synergistically. The court found that, read literally, these
statements expressed an aspiration/a reason for putting them together, not a
claim that they did work synergistically, especially since they were made
early on in the creation process. True, a reasonable reader might take away the
implication that the design worked, but there was no evidence of consumer
reception, so ChromaDex didn’t meet its burden. An internal email stating that in
a communication with a news outlet Elysium should “harp on the fact that the
synergistic effect of the combination is a huge benefit to consumers and a big
differentiator” wasn’t enough in the absence of evidence that the message ever
reached consumers.
Elysium also made statements about its “rigorous” safety
studies for NDI submissions and its compliance with FDA regulations/“Exceeds
FDA Recommendations.” Was this literally false because there was no NDI
submission? Elysium submitted evidence that it did conduct “rigorous” safety
studies, initially aiming for an NDI submission, and that when it used NR
supplied by ChromaDex for Basis, that ingredient did have NDI status. The
transition to IND status wasn’t shown to be based on any difficulty with the
NDI application, and it removed the reference after it decided to apply for IND
status. The statement didn’t necessarily or unambiguously convey that Elysium had
submitted an NDI notification; summary judgment was appropriate. Similar
reasoning applied to statements about licensing a pending patent (application).
Statements that allegedly falsely implied that Elysium was
significantly involved with the research behind the ingredients in Basis:
ChromaDex tested “[i]nside this bottle is 25 years of research,” and found that
“82.7% [of test respondents] answered that the materials communicate or imply
that the company conducted 25 years of research on aging, compared with 43.8%
for the control.” The control statement tested was: “Inside this bottle is
years of research, that was conducted by us and others.” [Note how badly that
control does: it is not effective at communicating its explicit message. I have
occasionally suggested that if you can’t get a control to avoid deception of a
substantial percentage of consumers, then it might not be worth requiring a
change unless the entirety of the message is not worth delivering to
consumers.]
The statements weren’t literally false claims that Elysium
itself was involved with all of the research. And the survey was specific to 25
years of research on aging, not anything else; ChromaDex didn’t show
that was false. “Basis is revolutionary because it’s the first product to come
out of really good aging research” was puffery as to “revolutionary,” and not shown
false as to “first.”
Likewise, statements that Elysium “works in partnership with
the world’s leading scientists and research universities,” and that their
“board guides the scientific direction of Elysium” were not literally false
claims that the advisory board “was itself involved in the research and
development or would vouch for Basis’s safety and efficacy.” “Absent a false
statement about what the advisory board has done or the capacity in which its
members serve, a company is not required to convert every advisor to an officer
or to follow each piece of advice it receives at the expense—if it does not do
so—of inviting a Lanham Act lawsuit.” And ChromaDex didn’t provide evidence
that consumers received the allegedly false message.
Elysium also showcases client testimonials on its website, e.g.,“I
liked the fact that Elysium has scientists on staff. I liked the fact that there is data …. The
fact that there are so many knowledgeable people involved in this, and that you
were so responsive when I had questions. It left me feeling secure in the
knowledge that I was given and made it worth a shot” and “I said, let me look
at this because of the credibility of the founder and his work at MIT. I was
buoyed by the idea that these people had confidence in the research and were
promoting it.” There was no evidence that these testimonials were fake or conveyed
implied falsehoods. And reasonable readers would take these as opinions. “[S]imply
indicating that a particular consumer was satisfied with a service plainly does
not constitute a false or misleading statement.” [Note that they would not
count as substantiation on their own.]
Elysium also counterclaimed, and ChromaDex kicked out some
of them while Elysium got partial summary judgment on the falsity of a
statement that ChromaDex was the only seller of NR.
ChromaDex’s website said that “[i]n 2004, Charles Brenner
PhD discovered a unique and overlooked form of vitamin B3 (nicotinamide
riboside) that is a natural precursor to NAD” and “Tru Niagen is a specialized
form of vitamin B3 discovered by our Chief Scientific Advisor Charles Brenner,
PhD and developed specifically to increase NAD more effectively than any other
B3 before it.” As to the first
statement, other material on the same page allegedly clarified that NR was identified
in the 1940s. But the court held that “the FAQs as a whole do not qualify or
dispel the notion that Brenner was the one who discovered NR and that he did so
in 2004.” Interestingly, the court rejected Elysium’s argument that the supposed
clarification wasn’t effective because it was in a different FAQ answer requiring
a different consumer click. This didn’t require a protracted “investigation
process.” [I don’t really understand the court’s position here. If the FAQ was
click to expand, then consumers would have to somehow figure out that
the answer to the sixth question was qualified by the answer to the 8th.]
Anyway, the statement “means, unambiguously and in plain
English, that Brenner discovered NR, which is a natural precursor to NAD, not
that he discovered that NR is a natural precursor to NAD.” The “context” of the additional statement that
“nicotinamide riboside was first identified in the 1940s” didn’t clarify what
the earlier statement meant; “it contradicts the earlier statement on its face
and, if credited, reveals that the earlier statement is false.” So too with
statement #2. This statement was not ambiguous. (However, the court refused to
consider other explicit variants of the statement, since Elysium didn’t identify
them in its complaint. Other variants might be relevant to scope of relief but
couldn’t be relied on to avoid summary judgment.) “[F]or a statement to be
ambiguous, requiring extrinsic evidence of meaning, there must be more than one
reasonable reading of the statement.” That wasn’t true here: the statement
plainly and unambiguously referred to NR, not synthetic NR or Niagen or Tru
Niagen, which were products and not molecules/things that could be discovered. It
was unreasonable to read this as a statement about synthetic NR, because that
would have to be created and not discovered.
ChromaDex’s statements that it “pioneered NAD research by
investing millions of dollars in safety and human clinical trials on its patent
protected NR” that it supplied NR to “over 160 leading institutions for
research” weren’t shown to be false despite Elysium’s argument that they created
the misleading impression that ChromaDex funded those studies; its evidence
didn’t show falsity as to ChromaDex’s research investments as a whole.
Allegedly false claims that Tru Niagen increases NAD+ levels
by 40–50% (qualified by “on average at 300 mg / day for 8 weeks”): There was a material
issue of fact as to whether the relevant study was manipulated/biased and thus
not capable of supporting these establishment claims.
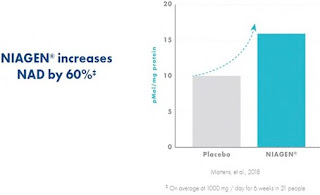 |
| increases by 60% graph with disclosure right underneath |
However, the claim that the product “increases NAD by 60%,” with a citation to a different study and the qualifier that this is “[o]n average of 1000 mg / day for 6 weeks in 21 people,” was not shown to be falsifiable; the qualifier wasn’t so “inconspicuously located or in such fine print that readers tend to overlook it” but was instead “the only other text on the graphic, displayed immediately below the graphic itself.”
For another study-related claim, “Niagen was
clinically-studied at 300 mg to increase NAD,” that didn’t explicitly claim that
the increase was statistically significant; if the press release containing the
claim had asserted that “Niagen was therefore effective, or something else
along those lines,” lack of statistical significance might matter, but it didn’t.
Safety/ “trusted” claims: Elysium alleged that Tru Niagen
could injure consumers by dramatically decreasing their white blood cell count.
The relevant study reports a statistically significant decrease in the white
blood cell count of patients taking 300mg of NR (the dosage of Tru Niagen)
compared to placebo. A jury could find a potential relationship between Niagen
and white blood cell counts, but there wasn’t enough evidence that the decrease
was enough to put consumers below the “normal” range or was otherwise unsafe.
Allegations that Tru Niagen falsely claimed safety/efficacy
review by the FDA, when GRAS/NDI submissions only relate to safety, and they
only related to Niagen and not Tru Niagen. Historical versions of ChromaDex’s
claims did have the first problem (e.g., “rigorously tested for safety and
efficacy with the US FDA”). What about current ads, e.g., “3 FDA Safety Reviews,”
that didn’t explicitly mention efficacy? Its survey expert found “a net 23.3%
of respondents who take away the mistaken belief that the FDA has reviewed Tru
Niagen for effectiveness.”
Representations in advertising that are “commensurate with
information in an FDA label generally cannot form the basis for Lanham Act
liability” unless an ad substantially deviates from the label. The same was true here. “If the challenged
statements regarding GRAS and FDA review for safety could have led consumers to
any confusion that the FDA also reviewed the product for efficacy, such
confusion could have been based only in confusion about the FDA and what it
means for the FDA to review a nutritional product.” That wasn’t ChromaDex’s
fault [so ChromaDex is allowed to exploit the misunderstanding], and so the court
wouldn’t “chill” speech informing the public about FDA review/notification. [It
doesn’t seem to be chilling to me to require companies to provide information
necessary to understand the claim.]
Statements that ChromaDex is the “only” source of NR: “NR is
a patented ingredient, only sold as NIAGEN. ChromaDex holds the patent rights
to NR, and sells the ingredient to consumers as TRU NIAGEN” and “Any
nicotinamide riboside product that does not say ‘NIAGEN’ on its label does not
contain nicotinamide riboside that has been successfully notified to the FDA.” But
ChromaDex does not own a patent on NR, a substance found in nature; it licenses
a patent for one method of manufacturing NR. The ad was not ambiguous: it
stated that only ChromaDex could sell “NR” and that only Niagen had NR. Elysium
was entitled to summary judgment on falsity here.
Disparaging statements: Elysium alleged that ChromaDex’s
warning against counterfeits attacked Elysium (and any other source of NR),
targeting it by intentionally using an image similar to Basis’s bottle on the
website. The webpage said, inter alia: “How
to tell if your nicotinamide riboside (NR) supplement is authentic, and why you
should know whether your product is counterfeit.” It also specifically targeted
Elysium’s “misleading marketing materials,” accusing it of using a different
ingredient than what it used to claim scientific support, “and there is no
published, peer-reviewed human data on ‘NR-E’ nor has it been reviewed by any
regulatory bodies,” in contrast to ChromaDex’s GRAS/NDI notifications (and other
positive things about ChromaDex).
 |
| authentic/safe/effective graphic |
“The record is undisputed that ChromaDex intended to communicate the message that Basis was inauthentic, unsafe, and ineffective.” However, like most Lanham Act courts, this one doesn’t change the standard for intentionally implied messages. The webpage didn’t explicitly say that Basis was inauthentic and counterfeit, unsafe, or ineffective, nor convey those things by necessary implication. A statement made by “clever innuendo” requires evidence of consumer reception, which Elysium didn’t provide. [Periodic reminder that the explicit/implicit distinction, as modified by puffery and falsity by necessary implication, stems entirely from judicial innovation rather than the language of the Lanham Act; it would be completely possible to recognize intentionally implied messages as so likely to be received that no additional extrinsic evidence of reception is required, the way that the FTC does and that courts presume consumer confusion from intent to confuse in trademark law, the other half of the Lanham Act—often on much worse evidence of intent than available in false advertising cases.]
Materiality: Here, the court concludes that extrinsic
evidence of materiality is not separately required. The factfinder “may rely on
the nature and content of the statement alone to satisfy its burden.” The
Second Circuit has said that “in many cases the evidence and the findings by
the court that a plaintiff has been injured or is likely to suffer injury will
satisfy the materiality standard—especially where the defendant and plaintiff
are competitors in the same market and the falsity of the defendant’s
advertising is likely to lead consumers to prefer the defendant’s product over
the plaintiff’s.” It relied on evidence of lost sales, which wasn’t present
here, but that case still supported the conclusion that extrinsic evidence is
not strictly required, because the court emphasized the common-sense centrality
of the falsely advertised product feature (ability to detect pregnancy duration
for a pregnancy test, in that case) and the parties’ direct competition to find
that the lost sales were connected to the false advertising.
In some cases, no purpose is served by requiring extrinsic
evidence of materiality where the claims are obviously material, like whether a
vitamin pill would kill you or how long you have been pregnant.
Thus, the court denied the parties’ cross motions for
summary judgment on the claims against them as to materiality. What about the
counterclaims relating to the “counterfeit” webpage? While it was “entirely
reasonable” to find the alleged misrepresentations material, Elysium wasn’t
entitled to summary judgment in its favor on that element.
Injury: Elysium got summary judgment on injury as to
ChromaDex’s claims against it. ChromaDex’s only proof of injury was its
excluded damages expert report, which simply assumed that all of Elysium’s
sales would have gone to ChromaDex without the allegedly false advertising. “[T]here
is uncontradicted evidence that Elysium made sales of Basis prior to any of the
allegedly false statements and there is also uncontradicted evidence that
Elysium has competitors in the marketplace other than ChromaDex.” The report
didn’t prove causation.
In a previous case, the Second Circuit held that a
presumption of injury would arise in a Lanham Act false advertising case where
each of two conditions was satisfied (1) the “plaintiff has met its burden of
proving deliberate deception”; and (2) the relevant market is a two-player
market. But ChromaDex didn’t show either of those things. Even if a jury could
find that every other competitor to Elysium used NR purchased from ChromaDex,
the court thought that accepting its claim would be inconsistent with Lexmark.
I find that a bit weird—the court was concerned that ChromaDex would be able to
win its claim “even if Elysium’s advertising did not cause any customer to
purchase a product from Elysium rather than from ChromaDex and even if
Elysium’s advertising caused no injury to ChromaDex’s reputation” because
ChromaDex’s customers lost out. But (1) materiality would fix that and (2) here
the alleged causation mechanism is precisely deception about the thing that
ChromaDex supplies to those customers, so even if consumers don’t know its name,
its reputation is what’s at issue. Lexmark, after all, involved a
supplier who allegedly was the only non-defendant supplier of the relevant product
components. But it was possible that only ChromaDex’s customers were injured by
the false advertising, and ChromaDex didn’t provide evidence of cognizable
injury.
Nor would ChromaDex be allowed to pursue disgorgement or
injunctive relief as an alternative, because actual injury/causation was an
element of the claim, so there could be no liability in the first place.
Damages and Elysium’s counterclaims: Elysium was seeking
only injunctive relief and disgorgement, and there were no arguments that it
couldn’t do that. Also, for the counterfeit page that referred to Basis by
name, it was entitled to rely on a presumption of injury from explicit
comparative advertising, and so it got summary judgment on this element.






No comments:
Post a Comment