Penn Engineering & Mfg. Corp. v. Peninsula Components, Inc., 2022 WL 3647817, No. 19-513 (E.D. Pa. Aug. 24, 2022)
This seems like a silly result to me, shifting the burden to comparative advertisers, but it's often much harder to get summary judgment in a trademark case than in comparable cases.
The parties compete in the industrial fastener market. PennEngineering
claims a PEM family of marks and sued Peninsula for trademark infringement,
counterfeiting, false advertising, and unfair competition.
The court denied Peninsula’s motion for summary judgment except
for pure keyword buys, counterfeiting (based on resales of PEM goods), and
claims based on marks Peninsula didn’t use. That left for trial claims based on
some online ads that used PEM in text and on cross-reference charts used for
comparison, as well as trade dress claims based on a “square-in-square” clinching
nut.
 |
| keyword ads, no use in text |
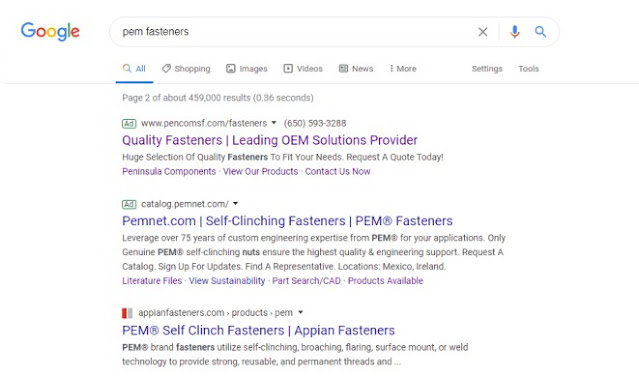
Bizarrely calling keyword buys “keyword conquesting,” PennEngineering sought summary judgment based on alleged sales diversion. The court pointed out that “diverting customers is a key aspect of competition. … Here, there is no dispute that the links are clearly labeled as belonging to Peninsula and there is no likelihood of confusion where the use of trademarks as trigger words is hidden from the consumer.”
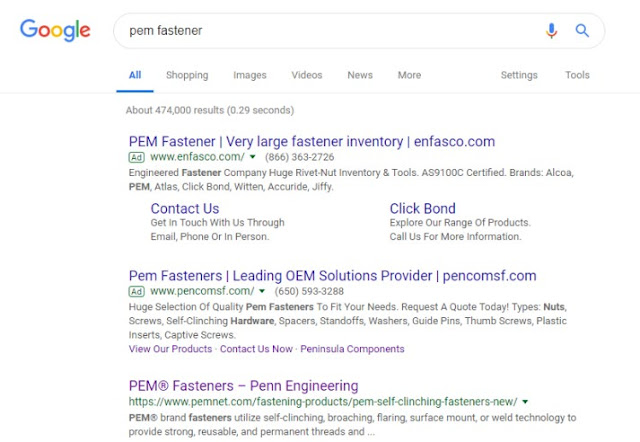
However, allegedly according to vendor mistake, some ads did show PEM in display text. The court found that the requisite intent is intent to confuse, not intent to use, and that a factfinder would have to decide whether these ads were confusing. “[A] reasonable jury could conclude that there is a likelihood of initial interest confusion from the visible use of ‘PEM’ in Peninsula’s advertisements because the advertisement is not clearly labeled. On the other hand, a reasonable jury weighing the Lapp factors could instead conclude that that there was no intent to confuse (rather, a vendor mistake) and that the average Internet user’s ability to understand the difference between ads and direct search results diminishes the likelihood of confusion in these limited instances.”
 |
| keyword ads with mark in text |
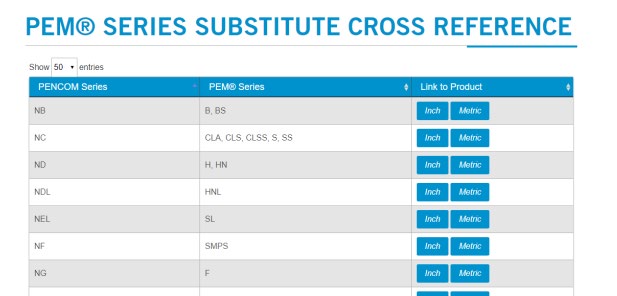
Because nominative fair use is the defendant’s burden in the Third Circuit, the court let claims based on cross-reference charts continue, even though it seems hard to understand why these comparisons would be confusing about source. Peninsula’s charts “identify Peninsula products that are available as substitutes for [PennEngineering’s] products.”
 |
| cross-reference chart |
Although the customers are sophisticated, the fasteners cost only pennies per unit; the parties also disputed whether “a handful of emails sent to Peninsula—which PennEngineering interprets as attempting to reach PennEngineering instead—constitute more than de minimis evidence of actual confusion.” So there might have been confusion, though the court didn’t explain why there’d be confusion because of the comparison charts.
The court said that it could be necessary to use the
plaintiff’s mark for comparison purposes even if fasteners are “staple items”
(commodities). [Notably, plaintiff apparently has two-thirds of the relevant
market.] “[I]t is clear that a party may use comparative advertising for any
items, including ‘staple’ items.” Although the court rejected the claim that a
cross-reference chart comparing types of Bob’s Burgers to McDonald’s burgers
would constitute trademark infringement, because “there is no likelihood of
customers confusing Bob’s Burgers and McDonald’s from looking at such a chart,”
the charts here were apparently different.
The strongest argument I can find for this result is plaintiff’s
argument that “the absence of disclaimers on the cross-reference chart ‘suggests
a manufacturer-distributor relationship, or that [Peninsula] is a division of
PennEngineering or a division of a common parent.’” Why would the relevant
consumers think that? What makes Bob's Burgers different? The court thought that this constituted a factual dispute
over whether the chart portrayed the true relationship between the parties.
The court also declined to grant summary judgment on whether
the claimed double square trade dress, which was registered, was functional—the
registration meant the burden was on defendant, and while its evidence created
a factual dispute, the registration also weighed in favor of nonfunctionality.
 |
| the design |
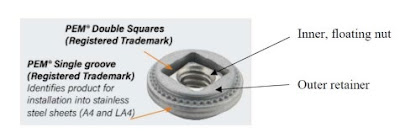
Peninsula, like PennEngineering, makes floating fasteners with square-in-square designs.
A floating fastener uses an outer
retainer that allows an interior nut some “float” to accommodate minor
misalignment. “Double squares” refers to a fastener with an interior square
that rotates with a screw and is stopped by an exterior square, within a
certain amount of “float” between the outer square’s corners for misaligned
parts, as shown below:
PennEngineering’s VP of Product Development testified about
the way the smaller square provides some float while the larger square prevents
rotation, which is “necessary for the functionality of the part.” PennEngineering’s
expert also opined that the interaction between the two square shapes “is a
functional feature of” the product. And Peninsula cited a prior trademark
prosecution by PennEngineering for admissions that the product design is
functional.
The Third Circuit very clearly doesn’t require that a
feature be “essential” to be functional. Nor did the existence of other designs
disprove functionality. “Further, PennEngineering does not show that these
alternative shapes would allow the same amount of float in both directions.”
Nonetheless, Peninsula didn’t meet its burden of proving
functionality, because it didn’t introduce its own expert report or explain
exactly how the design “affects the cost or quality of the article,” or how “the
‘exclusive use of [the feature] would put competitors at a significant
non-reputation-related disadvantage.’ ” PennEngineering’s expert opined that
the method of manufacturing the shape “entails the same cost and produces the
same quality for any shape, although ‘some shapes may be marginally easier to
form than others.’” That was enough to avoid summary judgment.
Counterfeiting through reselling legitimate PennEngineering
products: Protected by first sale. There’s an exception when goods are “materially
different,” which [supposedly, though not always in practice] requires
consideration of “whether the allegedly infringing products are likely to
injure the goodwill developed by the trademark owner in the trademarked goods.”
PennEngineering argued that resold products were “materially
different” because they weren’t covered by PennEngineering’s warranty or
customer service. But this wasn’t a material difference where no reasonable
person would ever claim on a warranty for a two-cent fastener (the warranty did
not cover consequential damages), and where its own VP of quality was unaware
of any warranty claims from reseller sales and even stated that, if such a
claim did occur, PennEngineering would likely honor its warranty the first time
for a given customer. PennEngineering also apparently was unpersuasive in analogizing
to an unauthorized used car dealer selling a Ford truck.
Such a truck would not have the
original Ford warranty and dealer services but would have the Ford logo and all
original parts without physical alteration. According to PennEngineering’s
theory of counterfeiting, the truck is “not a Ford.” Yet, a material
differences claim requires showing “differences that are likely to damage the
goodwill developed by the trademark owner.” Unlike a fake Gucci bag that falls
apart and injures an unsavvy, label-anxious customer’s goodwill toward Gucci, a
used Ford truck is still a Ford. PennEngineering’s “ceci n’est pas une Ford”
theory of counterfeiting falls flat.
False advertising: PennEngineering argued that Peninsula’s
use of cross-reference charts and copied performance data constituted false
advertising, because PennEngineering had test results supporting its
performance data and Peninsula’s comparisons were based only on its
expectations that the results would be the same.
The court declined to grant summary judgment on literal falsity.
In the Third Circuit, “although the plaintiff normally has the burden to
demonstrate that the defendant’s advertising claim is false, a court may find
that a completely unsubstantiated advertising claim by the defendant is per se
false without additional evidence from the plaintiff to that effect.”
So, were these claims completely unsubstantiated? “Peninsula
admitted that it reverse-engineers the parts and then assumes that the
performance ‘should’ be the same as the PennEngineering products.” By contrast,
“PennEngineering describes a time-consuming and expensive process to test each
of its products for performance ratings (e.g., how much weight each fastener
can secure).” Peninsula “publishes performance data on thousands of products
that show identical performance but … only independently tested around 51 of
its products,” and PennEngineering argued that it “sometimes disregards its own
test results to publish PennEngineering’s data instead.”
PennEngineering’s expert opined that the way in which the
fasteners are manufactured affects the final performance properties. Peninsula’s
Director of Product Development admitted that Peninsula does not know how
PennEngineering manufactures each part and does not specify manufacturing
processes for its manufacturing vendors.
However, PennEngineering did not actually show that any
particular performance rating published by Peninsula was incorrect.
For summary judgment purposes, Peninsula’s testimony that the
goal of the reverse-engineering program was to follow the “form and function”
to achieve the same performance, plus its expert test results showing that the
performance characteristics of a subset of the products tested were the same, would
allow a reasonable jury to conclude that Peninsula’s performance data has a
“semblance of support,” which would defeat the per se falsity theory.
PennEngineering also failed to show actual deception, as required because it
was seeking both injunctive relief and damages.
Likewise, the court denied summary judgment on the argument
that the charts would mislead consumers based on an expert’s opinion
(apparently not backed by a survey) that the cross-reference charts were likely
to mislead customers “into believing that the corresponding [Peninsula]
products are equal in quality and all other respects.”





