In re Kind LLC “Healthy and All Natural” Litig., 2022 WL
4125065, 15-MD-2645 (NRB), 15-MC-2645 (NRB) (S.D.N.Y. Sept. 9, 2022)
Plaintiffs alleged that KIND products displaying an “All
Natural/Non GMO” label were deceptive or misleading. Previously, the court
allowed NY, Florida, and California classes to proceed. But, because they
abandoned any claims based on the non-GMO part, the court found that they
lacked a viable claim that “All Natural” was misleading and decertified the
classes.
In 2015, FDA issued a warning letter to Kind about its
“healthy and tasty” claims, stating that the language was an “implied nutrient
content claim” and that certain KIND products did not meet the FDA’s saturated
fat content requirements necessary to describe food as “healthy.” “In response,
KIND argued that many universally recognized healthy foods such as almonds,
avocados, or salmon contain saturated-fat levels exceeding [FDA’s] limits.”
 |
| All Natural/Non GMO label, L, and revised Non GMO only label |
The resulting lawsuit was paused because the FDA pretended that it might actually do something about “natural” represenations. It didn’t, so eventually the case was unpaused. Other developments “sharply contracted” the scope of the claims to the “All Natural” claim on three product lines.
For all the claims, an objective reasonable consumer
standard applied. The court adopted a relatively new proposition—imported from
the Lanham Act and not traditionally part of consumer protection cases—that
“[t]o satisfy the reasonable consumer standard, a plaintiff must adduce
extrinsic evidence—ordinarily in the form of a survey—to show how reasonable
consumers interpret the challenged claims.” Thus, the claims required evidence
showing a reasonable consumer’s understanding of “All Natural” plus evidence
that the Kind products fell outside that understanding. Plaintiffs failed at
both points.
The earlier definition used in the case was heavily
dependent on now-abandoned “Non GMO” claims. “Non GMO” might give context to
the “All Natural” right in front of it on the label, but now there’s no falsity
claim about that. Without the GMO context, the court found that there was no
objective definition of “all natural.” FDA has some guidance, but its
application would depend here on what a consumer would expect to be in the
food, which is precisely what’s at issue. Plaintiffs’ own statements offered a
variety of understandings, which if not inconsistent were not all coextensive:
“that is, a product can meet the criteria in the FDA guidance that it does not
contain unexpected artificial ingredients without meeting the criteria in the
dictionary definition proffered by plaintiffs that it is ‘existing in or caused
by nature; not made or caused by humankind,’” and so on. “Given this diversity
of views, none of these definitions supplies, or purports to be, a reasonable
consumer’s definition of ‘All Natural.’” [Note: That’s not logically true: each
could be a reasonable consumer’s definition, but reasonable consumers could be
all over the map. For completeness, it would be useful to ask whether, given
all these definitions, Kind didn’t qualify according to a substantial
number/percentage of reasonable consumers.]
Nor did plaintiffs’ survey report provide a way to define a
reasonable consumer’s understanding of “All Natural,” because the court
excluded it.
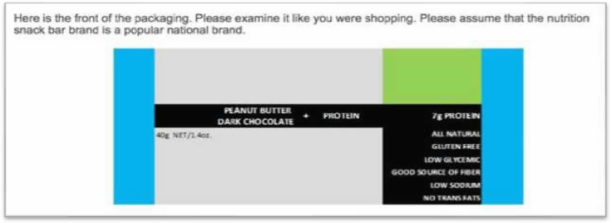
The survey presented individuals with “a mock-up of a
product, that, in many respects, resembled the packaging of a KIND bar.”
Note the omission of the non-GMO representation.
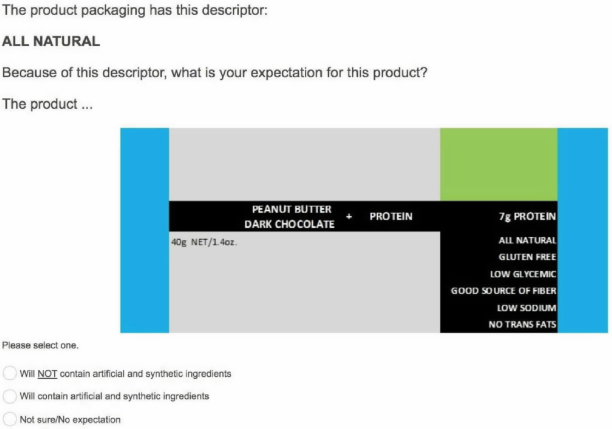
Then it asked them about whether they agreed, disagreed, or
didn’t know/weren’t sure about whether “All Natural” products would contain “artificial
or synthetic ingredients.” The survey found that 86.4% of consumers said no.
The survey also asked consumers to select one of the following options regarding their expectations of an “All Natural” product: (1) that it is not “made using these chemicals: Phosphoric Acid, Hexane, Potassium Hydroxide, Ascorbic Acid”; (2) that it “is made using these chemicals: Phosphoric Acid, Hexane, Potassium Hydroxide, Ascorbic Acid”; or (3) that they were “Not sure/No expectation.” The court really didn’t like this question, considering it misleading (e.g., Vitamin C is another name for ascorbic acid; some of the chemicals aren’t in Kind bars) and found that the surveyor’s “decision to blindly include items listed by plaintiff’s counsel in the complaint, without any investigation or consideration of the appropriateness of those items, only underscores his survey’s lack of reliability.”
The court found the survey inadmissible because it was
biased and leading. The first question
asked only about one potential definition and only allowed participants to select
whether they agreed, disagreed, or didn’t have an expectation. There was no contrast
with other possible meanings, and there were no open-ended questions. At
deposition, he said he was “test[ing]” the plaintiffs’ theory of liability, but
the court interpreted his answers as showing that his questions were designed
to support that theory. Relatedly, he chose to display the “All Natural”
claim in isolation, rather than as part of the “All Natural/Non GMO” statement,
as it always appeared on KIND labels, out of concern that the two “would
interact.” This made his survey less relevant.
Likewise, his second question listed “chemicals” drawn from
plaintiffs’ complaint, “without personally reaching any understanding of what
those ‘chemicals’ were, or whether they were ingredients that cannot be
considered ‘All Natural.’” [If the court had liked the theory better, he
probably would have been able to rely on other experts/facts provided by the
client for these points.] The word “chemicals” was leading, given “the
common-sense intuition that, when prompted by the word ‘chemicals,’ consumers’
consideration of the listed substances described as chemicals is tainted by the
connotation that ‘chemicals’ carries.” The failure of the survey to define the
terms and including Vitamin C under the name “ascorbic acid” to parallel “phosphoric
acid” was “a clear attempt to manipulate consumers into selecting the answer
that plaintiffs preferred.”
Even without the leading questions, the court concluded that
the survey profided “no useful information” about how a reasonable consumer
understands “All Natural.” The survey didn’t define “artificial” or
“synthetic,” or what it means for a product to “contain” or be “made with”
those ingredients. So, for example, the survey required further inquiries:
• What processing, if any, does a
reasonable consumer believe can occur to an ingredient or product before that
ingredient or product is considered artificial or synthetic?
• Are ingredients that do occur
naturally, such as Vitamin A or C, but potentially manmade in the specific form
that appears in KIND products, artificial or synthetic?
• Are trace or residual amounts of
chemicals that were used in processing ingredients in KIND bars enough to cause
the KIND products to contain “artificial or synthetic ingredients”?
Here, the answers to those questions were “central” to
plaintiffs’ theory of falsity. But the survey left a factfinder guessing at
them.
Plaintiffs argued that they didn’t need “a universally
accepted definition of ‘All Natural’ ” as long as they showed that a reasonable
consumer would interpret the claim to mean that the product didn’t contain
those specific artificial and synthetic ingredients. But they didn’t show that;
all they showed was that, “when provided with the definition of ‘All Natural’
that plaintiffs’ counsel constructed for this litigation,” respondents would
click a check box saying that they agree to it. That wasn’t enough to show that
reasonable consumers would be deceived when they saw the label.
Contra the FTC’s position on claims like “environmentally
friendly,” the court further held: “Nor can defendant be held responsible for a
host of possible, even if potentially reasonable, consumer beliefs about the
meaning of ‘All Natural.’ Such multiplicity distorts the reasonable consumer
standard.”
Without expert testimony, plaintiffs failed to show how a
reasonable consumer would understand “All Natural” on Kind products. Internal
KIND documents, statements of KIND’s founder, and a survey referenced in the
FDA’s solicitation of comments regarding the “All Natural” claim were
insufficient substitutes. The internal statements “just represent the views of
KIND employees or internal KIND survey data,” not reasonable consumers’
beliefs. [I don’t understand why the internal survey data aren’t about consumer
beliefs.] And the FDA noted that “consumers regard many uses of this term as
non-informative.”
Even if the survey did establish a reasonable consumer’s
understanding, the plaintiffs failed to develop evidence that any KIND product
claiming to be “All Natural” contained “artificial or synthetic” ingredients or
any of the chemicals the survey listed, due to deficiencies in that area of
proof. To the extent that three ingredients were lab-made—Vitamin E acetate;
ascorbic acid (Vitamin C); and Vitamin A acetate—they were only present in a
bar that displays prominetly on the front of the packaging that it is a KIND
bar “plus” “50% DV Antioxidants,” namely, “Vitamins A, C, and E.” “As such, no
reasonable consumer could have been deceived by the addition of added vitamins….
No reasonable consumer could believe that they would receive 50% of their daily
value of vitamins from a single bar without an artificial or synthetic vitamin
being added to the product.”
 |
| Antioxidant bar |
Given all this, the court also decertified the class.






No comments:
Post a Comment