MacuHealth, LP v. Vision Elements, Inc., 2023 WL 3863341, No. 8:22-cv-199-VMC-JSS (M.D. Fla. Jun. 7, 2023)
MacuHealth sells a nutritional supplement of the same name
that is intended to maintain or improve eye health; Vision Elements is a
competitor that sells Early Defense. Each capsule of MacuHealth’s product
contains three active ingredients, LMZ carotenoids, and Vision Health states
that Early Defense has the same active ingredients in the same amounts. Approximately
1 percent of Early Defense product sales are direct to consumers and 99 percent
are to eyecare physicians.
 |
| MacuHealth bottle: note colors |
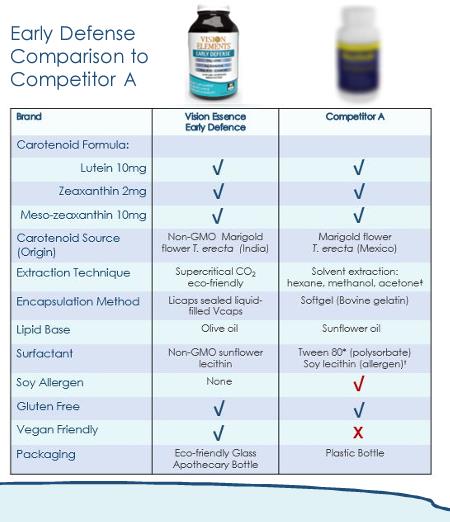
“The LMZ carotenoids used in MacuHealth and Early Defense are extracted and derived from marigold flower petals using solvents. It is industry standard to use as solvents hexane or methanol to extract and derive the LMZ carotenoids. The solvents are largely removed from the LMZ carotenoids during the production process, but residual amounts remain in the final product.”
Vision Elements’ challenged ads stated that Early Defense
had “Clean label ingredients: Solvent-free carotenoids derived from non-GMO
marigold flowers through an eco-friendly super critical CO2 extraction process
– No hexane, methanol, or acetone” and the bottle states that Early Defense
“contains none of the following:...Class 2 solvents – hexane, methanol,
acetone[.]” The FDA states that Class 2 solvents “should be limited in pharmaceutical
products because of their inherent toxicity.” These claims were repeated at
trade shows, in personal product pitches, and in emails to potential customers.
Vision Elements’ ads used comparisons based on the
MacuHealth bottle, labeled “Competitor A.”
Vision Elements also used “Competitor Solvent Extraction
Method” at trade shows, a document that includes a cover page of a patent
assigned to MacuHealth’s LMZ carotenoid supplier, and compares that patent’s
use of hexane as an extraction solvent to the use of supercritical CO2
extraction techniques. MacuHealth is the only competitor of Vision Elements
that used that supplier.
 |
| Vision Elements comparative ads |
Unfortunately for Vision Elements, it turned out that its own source also used hexane extraction. It didn’t inquire into the ultimate source or methods of production until the litigation began. Vision Elements initially relied on the certificates of analysis it received from its supplier to demonstrate that the supercritical CO2 extraction method was used on the batches it purchased, but ultimately admitted that the certificates did not provide a basis for determining whether Class 2 solvents were used. MacuHealth presented evidence of residual solvents in Early Defense capsules.Vision Elements chose not to test for the presence of solvents in Early Defense.
Vision Elements’ principal testified that the solvent claims
were “highly important” to customers and that it distinguished Early Defense
from competitors like MacuHealth.
An attendee at an industry conference asked, “How is it that
[MacuHealth] doesn’t have a way to do things without solvents?” One substantial
customer and reseller of MacuHealth for many years switched and promoted Early
Defense based in part on Vision Elements’ “clean formulation,” but, along with
another doctor who switched, stated in an affidavit that Vision Elements’
advertisements promoting Early Defense as free from Class 2 solvents did not
affect her decision to purchase Early Defense or recommend it to patients.
Unsurprisingly, there were no genuine disputes on literal
falsity; thus no additional evidence of consumer deception was required. There
were no genuine disputes on materiality either—the court pointed to Vision
Element’s own admission; potential consumers’ questions to MacuHealth; and the
“prevalence” of the solvent claims across Vision Elements’ website, its bottle,
and its oral and email pitches to potential consumers. “Through its own
actions, Vision Elements has demonstrated that its solvent claims are material.”
However, there was a genuine dispute of material fact on injury. There was evidence that customers switched due to “clean” claims, but their affidavits indicated that it wasn’t because of the solvents. (Yeah, I have to wonder if they’re worried about their own liability too.) The court declined to apply a lesser standard for injunctive relief—demonstrating again that the TMA’s presumption of irreparable harm only works for trademark claims, for which harm is not a recognized element of the cause of action; where harm has to be shown as part of the claim, the presumption doesn’t do any work. FWIW, I would have presumed harm from the evident materiality of the claim and the direct comparative advertisement, at least for injunctive relief.
This
also controlled the results for FDUTPA and common law unfair competition.
Negligent misrepresentation: “To state a cause of action for
negligent misrepresentation in Florida, a plaintiff must allege: ‘(1) the
defendant made a misrepresentation of material fact that [it] believed to be
true but which was in fact false; (2) the defendant was negligent in making the
statement because [it] should have known the representation was false; (3) the
defendant intended to induce the plaintiff to rely...on the misrepresentation;
and (4) injury resulted to the plaintiff acting in justifiable reliance upon
the misrepresentation.’ ” Here too, there was no genuine dispute on the first
three elements.
No comments:
Post a Comment